Recently, a breakthrough achieved by the teams of Professor Tong-Bu Lu and Professor Zi-You Yu from the Institute for New Energy Materials and Low-Carbon Technologies at our university, regarding the electrocatalytic reduction of CO2 to high‑concentration liquid products, has been accepted by the top international journal Nature Catalysis. The paper is titled “A high‑flux membrane electrode assembly for CO₂ electroreduction to 4.5 M formate with over 8,000 h stability”. The corresponding authors are Professor Tong-Bu Lu and Zi-You Yu, and the first author is PhD student Jing-Jing Li.
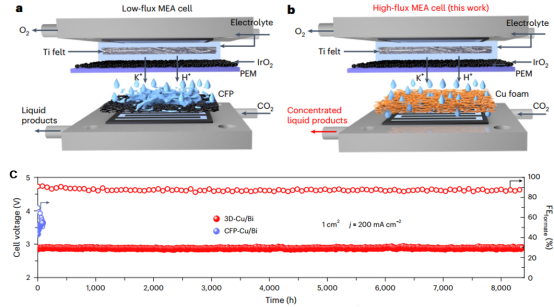
The use of renewable electricity to drive the electrocatalytic reduction of CO2 into value‑added chemicals represents an effective route toward carbon neutrality. However, the resulting liquid formate product is usually mixed with a large amount of electrolyte, leading to a low product concentration and significantly increasing the cost of downstream separation and purification. Although membrane electrode assemblies without catholyte enable the synthesis of high‑concentration formate, they generally suffer from operational lifetimes of less than 100 hours, a bottleneck that severely hinders their industrial application.
To address this challenge, the study proposes using high‑flux copper foam as the catalyst support. Its abundant and interconnected three‑dimensional macroporous structure provides efficient mass‑transfer channels for the liquid product, facilitating rapid desorption and directed transport of the product away from the catalyst surface. This allows continuous, long‑term production of highly concentrated formate. Using Cu/Bi core–shell nanowires as the catalyst, under conditions of feeding only dry CO2, no catholyte, and a current density of 200 mA cm-2, the electrolyzer continuously produces 4.5 M formate for over 8,000 hours with a Faradaic efficiency consistently around 90%. This performance represents the highest level reported in the literature to date. To further validate application potential, the electrolyzer was scaled up to an active area of 100 cm2, where the catalytic system retained excellent activity and stability, laying the foundation for future industrial scale‑up. A pilot‑scale test is currently underway in collaboration with PetroChina Lanzhou Petrochemical Company.
The acceptance of this work by Nature Catalysis once again marks a major breakthrough achieved by our faculty members and research teams in the fields of materials chemistry and new energy research.
Nature Catalysis is a premier sister journal of Nature, publishing important research across catalysis and energy. Its current impact factor is 44.6.